July 31, 2025
J&J’s blockbuster drug Stelara saw sales plunge 43% in Q2 2025, yet the company still delivered a solid overall performance driven by its Innovative Medicines segment, which crossed $15 billion in quarterly sales for the first time. Overall revenue reached $23.7 billion, representing a 5.8% year-over-year increase, while adjusted EPS was $2.77, surpassing Wall Street expectations and prompting management to raise full-year guidance to ~$93 billion.
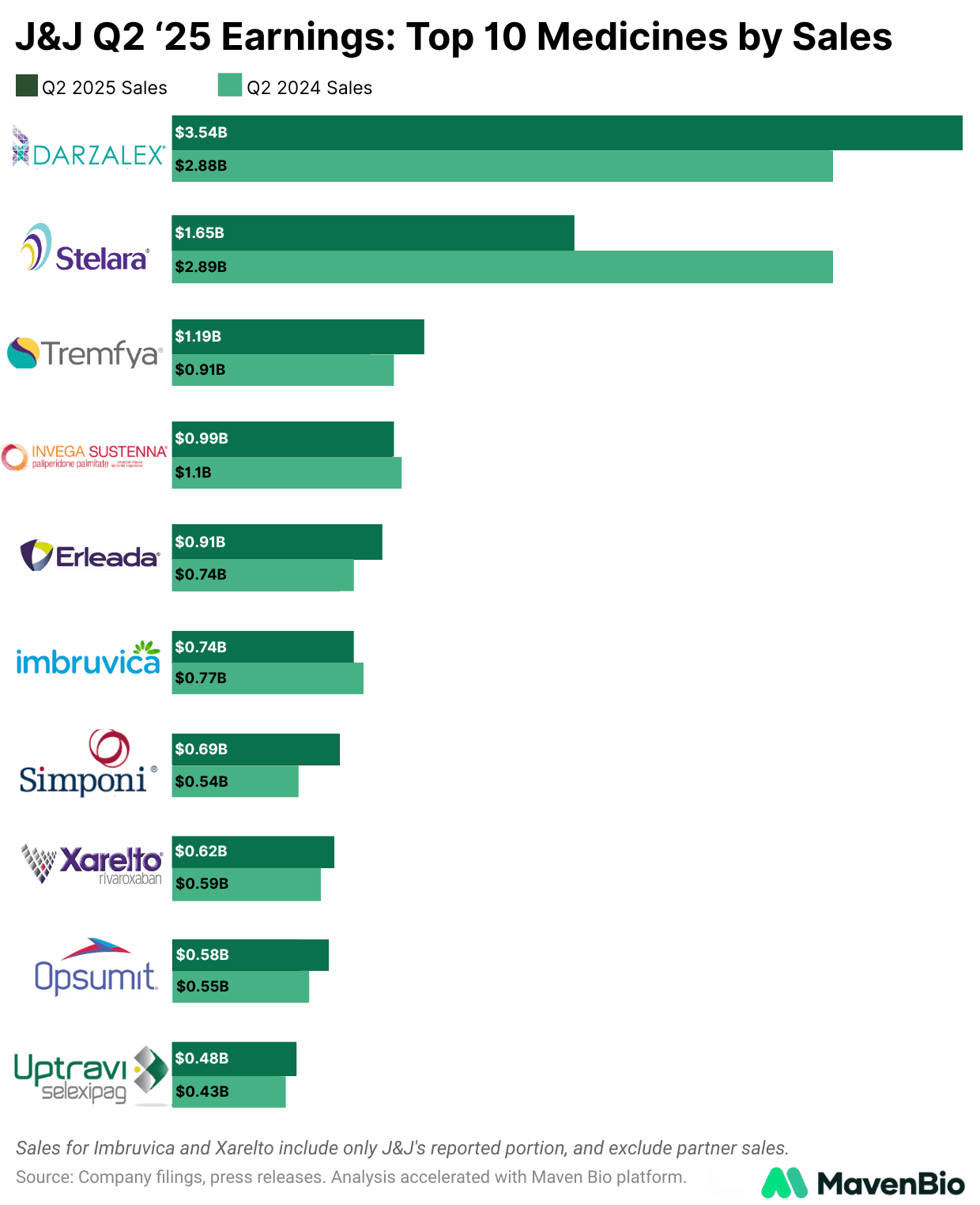
In oncology, Darzalex further cemented itself as the cornerstone of J&J’s portfolio, achieving sales of $3.54 billion, a notable 23% increase from the previous year. Growth was primarily driven by share gains of 4.1% across all treatment lines and nearly 8 points in frontline settings.
Moreover, Darzalex's potential FDA approval for treating high-risk smoldering multiple myeloma, following a favorable 6-2 ODAC vote in May, sets the stage for further expansion. With composition-of-matter patent protection extending to 2029 and strong adoption globally, Darzalex is well-positioned to sustain momentum in J&J’s oncology lineup.
Carvykti, J&J’s pioneering CAR-T cell therapy, also demonstrated exceptional performance with Q2 sales of $439 million, more than doubling year-over-year from $186 million. The recent five-year update from the CARTITUDE-1 trial, showcasing median overall survival exceeding 60 months from a single infusion, continues to validate Carvykti’s potential. Nonetheless, revenue acceleration remains somewhat constrained by production capacity limitations. J&J and partner Legend Biotech have responded by investing $150 million in expanding their manufacturing facility in Belgium.
The Rybrevant and Lazcluze combination therapy for EGFR-mutant lung cancer recorded substantial growth, generating $179 million this quarter. Recent compelling overall survival data favorably positions this combination against AstraZeneca’s established Tagrisso therapy, though the lung cancer market remains competitive. Meanwhile, Erleada achieved impressive sales of $908 million, up 23% year-over-year, demonstrating strength in prostate cancer treatment across metastatic and non-metastatic settings.
Blockbuster immunology drug Stelara faced a substantial 43% sales decline in Q2 2025, dropping to $1.65 billion amid intense competition after losing patent exclusivity. Multiple FDA-approved biosimilars, including Wezlana, Starjemza, and Selarsdi, have rapidly eroded Stelara's market share.
In immunology beyond Stelara, Tremfya emerged as a major growth driver, reaching $1.19 billion in sales, a robust 31% increase year-over-year. Expanded indications in psoriasis, psoriatic arthritis, ulcerative colitis, and Crohn’s disease, supported by positive long-term extension data from the QUASAR study (70% clinical remission and 43% endoscopic remission at 92 weeks), position Tremfya strongly toward achieving J&J’s $10 billion annual sales target.
Simponi, another immunology asset, also posted strong performance with a 29% increase to $690 million, driven by robust international demand. Neuroscience asset Spravato saw impressive growth of 53%, following FDA approval as the first monotherapy for treatment-resistant depression, which more than doubles its addressable patient population.
This quarter also featured significant pipeline advancements. FDA approval of Imaavy for generalized myasthenia gravis expanded J&J’s therapeutic scope into rare neuromuscular disorders. Furthermore, TAR-200 received priority review status for non-muscle-invasive bladder cancer after demonstrating impressive complete response rates in the SunRISe-1 Phase 2b trial, with a regulatory decision anticipated later this year.





